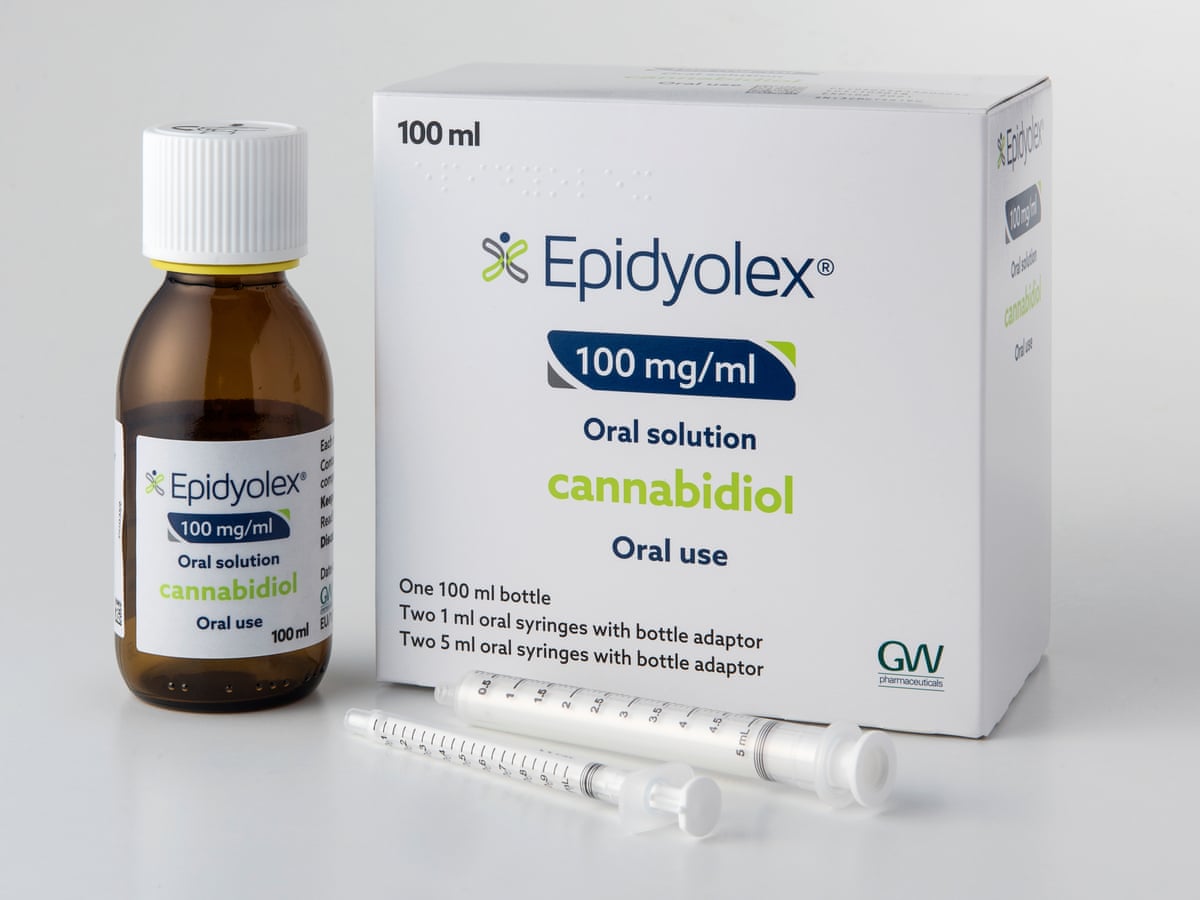
The National Institute of Health and Care Excellence (NICE) has said that they’ll not recommend cannabidiol (brand name Epidyolex) to treat TSC-related epilepsy in NHS England. We need YOU to help us change NICE’s mind.
The TSA is bitterly disappointed by NICE’s decision, with cannabidiol being an important option in treating and managing TSC-related epilepsy. Although NICE has come to the wrong conclusion, there’s still time to show them why it’s vital that cannabidiol is funded in England.
We believe that everyone affected by TSC deserves the opportunity to explore a range of treatment options, including cannabidiol when appropriate and recommended by a clinician. We’ve carefully read NICE’s reasons for their decision, and believe that these key areas have been overlooked:
- We know that a reduction in seizure frequency can have a massive positive impact on quality of life for people affected by TSC. Even if someone is not seizure free, having fewer, predictable seizures can really make a big difference
- The role of seizure reduction in challenges around care responsibilities is overlooked. Care for people with TSC (which is often at least two-to-one) can lead to secondary challenges in employment, financial security, social interactions and the wider family unit (such as the impact on siblings). Seizure reduction can play a major part in making these challenges easier
- There is currently an unacceptable inequality in access to TSC-related epilepsy medicines. Decision-makers in Wales and Scotland have already made correct choices to fund cannabidiol, which we campaigned for. That an important TSC-related epilepsy is available to some people but not others depending on where they live in the UK is completely intolerable for the TSA
What we need you to do
Share your views on the impact of living with TSC-related epilepsy in this survey (survey now closed). We’ll then share these views with NICE, to emphasise how important seizure reduction is on quality of life.
We want to emphasise to NICE the massive impact that cannabidiol can have on quality of life, in particular by reducing the number of TSC-related seizures. We feel that NICE’s decision has focused too much on being completely seizure free, instead of looking at the benefit on having fewer seizures.
However, the TSA’s views can only be impactful if people in the TSC community – the most important people in this decision, who face the impact of TSC every day – echo our opinions. This can be done by filling out our short survey.
Your local MP can have a big influence on changing NICE’s mind, such as by adding their voice to the campaign or by raising the topic in the House of Commons.
It takes just a few easy steps to contact your local MP:
- Find your local MP on the UK Parliament website
- Find your local MP’s email address or constituency postal address
- Download and use our template letter or email (or you could write something yourself)
- Update the TSA on any replies that you receive
Go that extra step: Contact Liz Twist (Chair of the All-Party Parliamentary Group on Rare, Genetic and Undiagnosed Conditions) and use this template (liz.twist.mp@parliament.uk / 0191 414 2488).
The steps taken by NICE to review funding requests for new medicines can be complicated. These are the key steps to a medicine review:
- Evidence is shared with NICE from different people to show why a medicine should be approved for funding. This includes representatives of people who will benefit from the medicine (such as the TSA and people with TSC), the company who is making the medicine, and medical experts
- NICE review the evidence that has been given to them and decide whether to say ‘Yes’ or ‘No’ to funding the medicine
- This is where we are now. If NICE don’t say ‘Yes’, they let everyone who submitted evidence know. There is then a short period of time for everyone who submitted evidence to try and change NICE’s mind. This is done by sharing more evidence or showing anything important that NICE didn’t focus enough on. NICE will then take the new evidence and re-review
- If NICE still say ‘No’, this is now considered their official view. However, there’s still the opportunity to appeal the decision. In the past, the TSA has helped to change NICE’s mind at this point, such as when everolimus (brand name Votubia) was initially rejected by NICE but then approved on appeal
Everyone who has TSC should have the opportunity to explore all medicines that could be of benefit, including cannabidiol. We can convince NICE on the benefits of cannabidiol only with your involvement. Help us to change the landscape of treating TSC-related epilepsy in England.
Make a one off or regular donation
£10 Can allow us to send a welcome pack to a family who has just received a life-changing TSC diagnosis, ensuring that they do not go through this time alone.
£25 Can help us develop materials that are included in our support services, flagship events or campaigns.
£50 Can provide laboratory equipment for a day’s research into the causes, symptoms, management or treatment of TSC.
To provide help for today and a cure for tomorrow